Document Control Software Overview
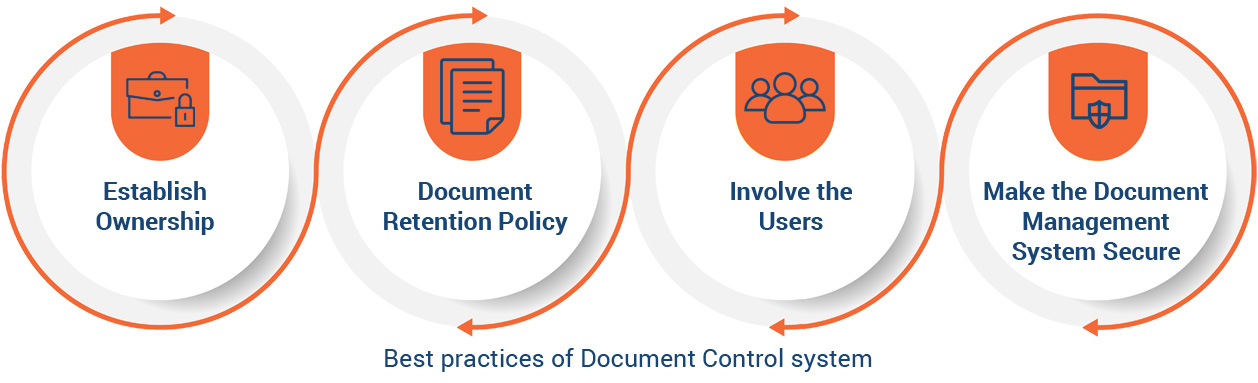
Documents are an essential part of every business and can take many forms. At any enterprise, documents can include contracts with suppliers, customers, employees, and guidelines for governance. Document Management and document control are absolutely critical from a quality management perspective. In fact, a robust, flexible, scalable document control software is the first step in the automation of quality processes and workflows using an EQMS.
Only with the right document control strategy can the quality processes within your organization be truly collaborative. It is at the core of compliance with FDA and ISO requirements, in addition to adherence to other compliance and regulatory requirements. Document control also plays an important in improving the efficiency of processes by enabling reliability and traceability of all quality control-related data and information.
In today’s digital era of quality management, a data-driven approach to building a quality culture within your enterprise is key. Your document control software is at the core of any data-driven EQMS system.
Request an Online Demo