Medical Device Quality Series: Reporting Adverse Events to the FDA with Ease – A CQ Guide
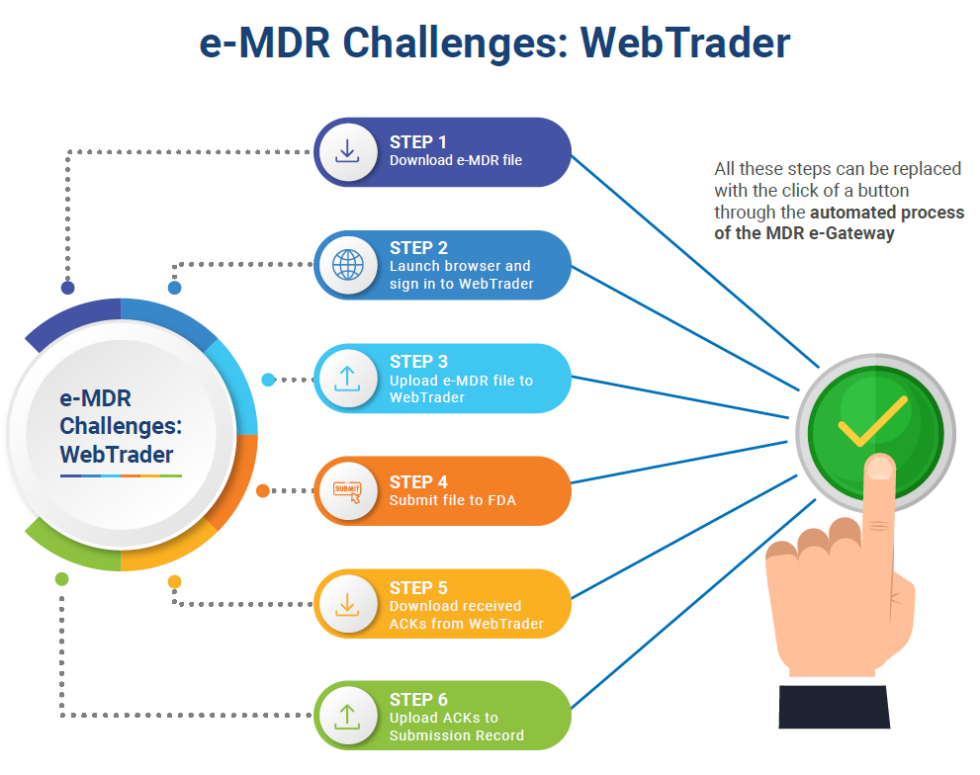
In one of our earlier CQ Guides, we spoke about the importance of garnering insights from complaints data to improve quality metrics in the medical device industry. In this post – as part of our Medical Device Quality series – we’ll talk about streamlining the process of reporting adverse events to the FDA, especially in scenarios where the no. of e-MDRs that get submitted is fairly high.